We use our unique microRNA-based biomarkers to personalize cancer treatment.

We partner with cancer clinics and pharmaceutical companies

Products
Learn about PROSTOXTM Test here, the first radiogenomics assay that can help prostate cancer patients avoid toxicity from radiation therapy.
MiraDx is developing more panels of biomarkers to apply before radiation therapy treatment to identify patients who have different sensitivities and responses to radiation therapy. These tests make much needed strides towards personalizing radiotherapy. More about our research on Radiogenomics
Recent data now indicates that changing estrogen levels, specifically estrogen withdrawal, can trigger cancer in women with the KRAS-variant. Knowing a woman’s KRAS-variant status is an additional piece of critically important information to help direct decisions for estrogen management, as well as primary and secondary cancer screening. More about KRAS-variant testing
MiraDx is creating biomarker panels to predict both toxicity (immune-related adverse events (iRAEs)) as well as response to the developing immunotherapies. Their first test predicts iRAEs to anti-PD1/PDL1 therapy across all cancer types. So, patients with any type of cancer who are being considered for this therapy are eligible for testing. More about PrevIOTOX testing
Featured Study
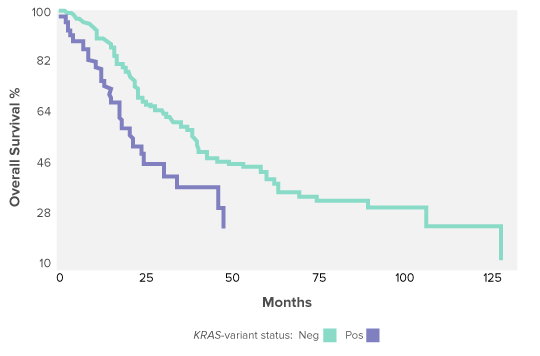
A KRAS-variant is a biomarker of poor outcome, platinum chemotherapy resistance and a potential target for therapy in ovarian cancer.
The KRAS-variant predicts significantly worse overall survival for post-menopausal ovarian cancer patients over 52 years of age. Overall survival for ovarian cancer patients with and without the KRAS-variant are compared using Kaplan Meier analysis. Outcome is significantly worse for KRAS-variant positive EOC patients over 52 years of age by log-rank test. This poor outcome is due to Platinum resistance for KRAS-variant patients which has been found across many cancer types.
Interested in partnering with us?
Contact Us
ADDRESSES
CLIA Lab Address
Administrative Address
Information
- Privacy Policy
- Financial Conflict of Interest Policy
- MiraDX CLIA certification #: 07D2006340



