PROSTOX is Clinically Validated to Help Physicians Identify Patients at Risk of Developing Significant Late Genitourinary (GU) Toxicity
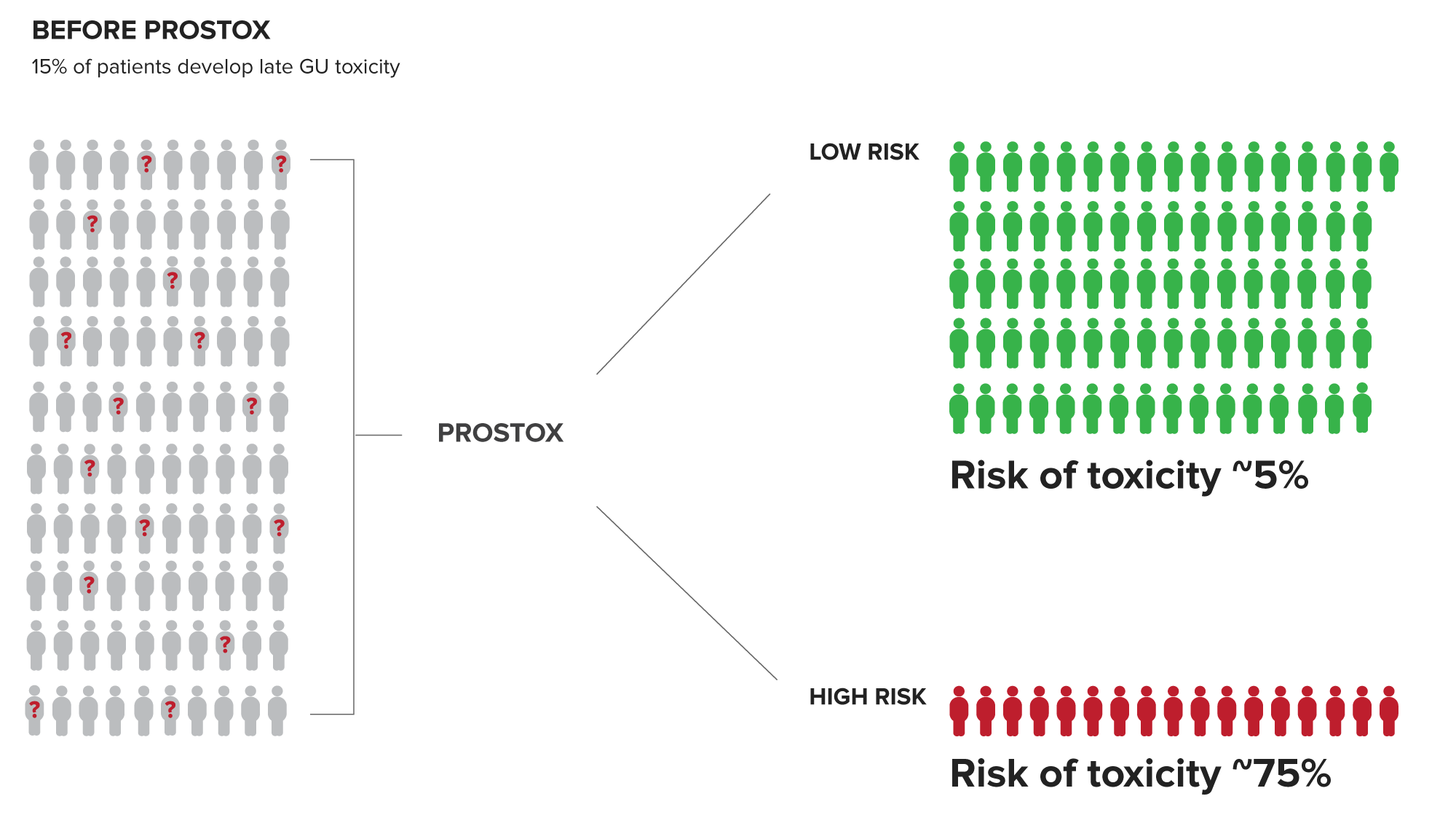
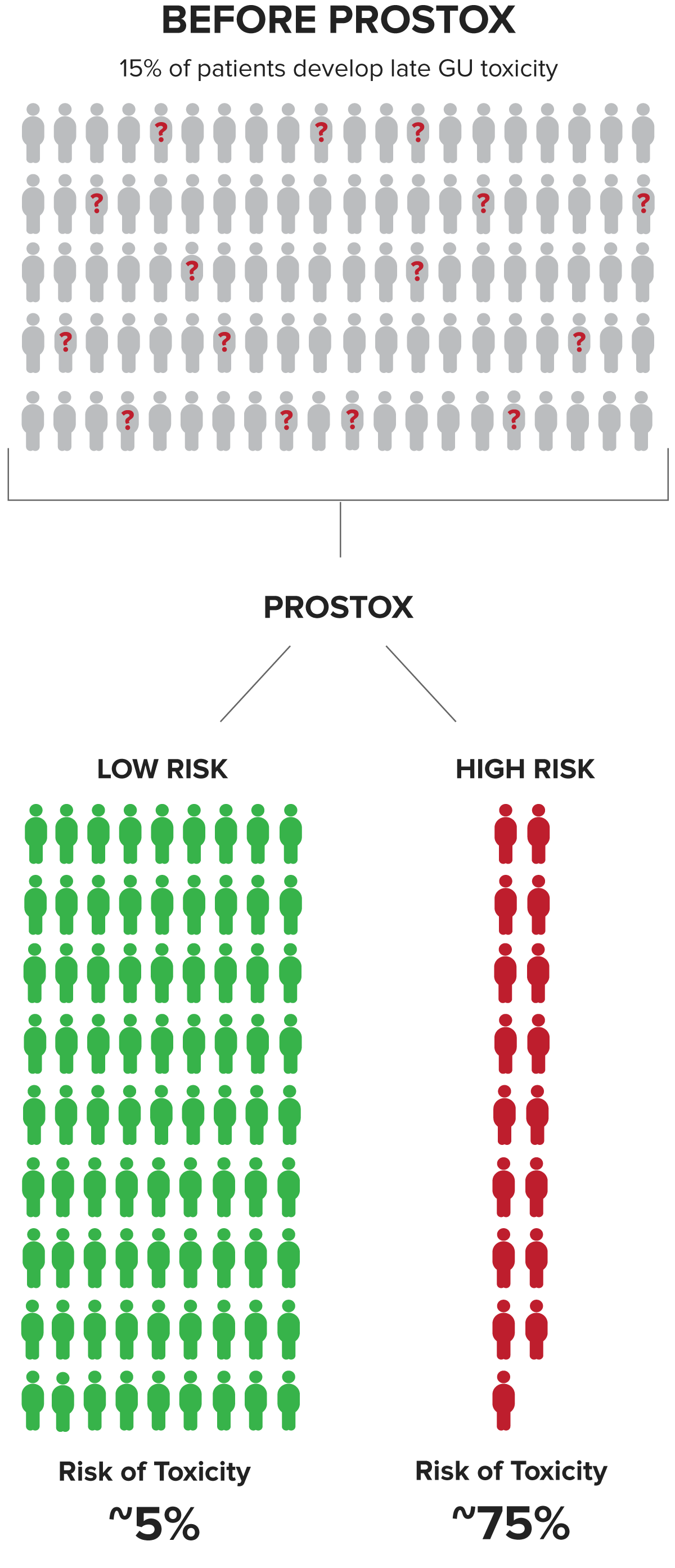
Patients at high risk have a ~75% chance of developing toxicity from SBRT or CFRT. This valuable insight enables the risk of toxicity to be part of the cancer treatment consideration.
Discover Precision, Clarity, and Confidence
Combined analysis of clinical validation and research studies accessing PROSTOX ultra performance.
94%
SPECIFICITY*
76%
PPV*
Positive Predictive Value
86%
NPV*
Negative Predictive Value
10Fold
LESS TOXICITY**
Late grade ≥ 2 GU
*Kishan et al. Clin Cancer Res 2025.
**Kishan et al. Radiother Oncol 2022.
Publications
MiraDx is paving the future of personalized medicine, enabling safer, more effective treatment decisions tailored to each patient’s unique genetic profile.
Explore our publications and learn how PROSTOX can transform your patients’ cancer care.
2025 | Clinical Validation
PROSTOX, a signature of late GU toxicity after SBRT radiotherapy in MIRAGE, a prospective trial
Phase III, randomized clinical study
PROSTOX can accurately predict late Grade ≥ 2 GU toxicity after SBRT, regardless of treatment modality.
See Publication2023 | Clinical Utility
The impact of a genetic signature of late radiation toxicity on prostate cancer treatment decision making
Phase II, single-center, prospective clinical study
PROSTOX predicts toxicity risk from SBRT or CFRT and influences treatment decisions for localized prostate cancer.
See Publication2023 | POST-OP Clinical Validation
Application of a genetic signature of late GU toxicity in SCIMITAR, a Post-op SBRT trial
Phase II, dual-center, single-arm clinical study
PROSTOX identifies a higher risk for late grade ≥ 2 genitourinary (GU) toxicity following post-prostatectomy SBRT.
See Publication2022 | Development Study
Germline variants disrupting microRNAs predict long-term GU toxicity after prostate cancer radiation
Developmental Study
Germline mirSNP-based predictive models effectively forecast late-grade ≥ 2 GU toxicity following CFRT or SBRT.
See PublicationPROSTOX
